Inside the Weird World of 3D Printed Body Parts
Backchannel

Startups in the U.S. are working on printing nipples and bits of liver tissue, while a Russian provocateur claims to have on-demand thyroids.
![]()
Laura Bosworth wants to 3D print breast nipples on demand. The CEO of the Texas startup TeVido Biodevices is betting on a future in which survivors of breast cancer who have undergone mastectomies will be able to order up new breasts printed from their own living cells.
“Everyone,” she says, “knows a woman who has had breast cancer.” Right now their options are limited. Reconstructed nipples using state-of-the-art plastic surgery techniques, she says, “tend to flatten and fade and don’t last very long.” A living nipple built from the patient’s own fat cells, and reconstructed to the precise specification of the original nipple, could go a long way to ameliorating the psychological trauma often associated with mastectomies.
Bosworth readily acknowledges that significant obstacles must be overcome before 3D printed breast parts become an affordable reality. Despite the waves of hype that surged after Anthony Atala, a Wake Forest professor, wowed a TED crowd in 2011 by purporting to print a human kidney on stage, no one has yet used a 3D printer to create a functional human organ.
The science is only half the battle. Venture capitalists aren’t exactly beating down the doors of TeVido. It’s a lot easier, observes Bosworth, to raise money “for an app that lets you order a taxi” than for a biomedical breakthrough that will cost millions of dollars in R&D before beginning the lengthy process of clinical trials needed to bring a product to market. Yet Bosworth is convinced that a $6 billion market awaits whoever gets out of the lab first. “The field itself has grown tremendously,” says TeVido co-founder Thomas Boland, one of the first scientists to start modifying ordinary 3D printers to print layers of living cells instead of ink. Researchers far afield, in China and Russia and Switzerland, at Ivy League labs and in the biotech hotbed of San Diego, are all pushing bioprinting forward. The disciplines of material science, cell biology and computer-controlled manufacturing are all merging.
If we believe everything we’ve heard recently, we’ll be 3D printing our food, our cars, our homes, our electronics—heck, the entire structure of globalized trade will be disrupted when we’re 3D printing everything we need in our living rooms rather than having it shipped in containers from China. The possibilities seem near infinite, even if the present-day realities are constrained.
As overblown as this swirling rhetoric may seem, the realms of science fiction and cold, hard bioprinting fact have convened at least once in real life, in the uncanny meeting of the minds of a nipple builder, a pioneer in tissue fabrication and a (possibly) mad Russian futurist. In that brief convergence one can glimpse the grandiose visions that cautious scientists tend to keep to themselves. Absurdity mingles with the commonplace. And you’re reminded of the most astounding thing of all: how today we’re talking, in matter-of-fact, business-savvy tones, about the actual printing of human body parts.
Now, back to the nipples.
![]()
In 2000 Thomas Boland was an assistant professor at Clemson University, in South Carolina, when he first conceived of modifying a standard HP inkjet printer to place layers of cells on top of one another, earning him the sobriquet “the grandfather of bioprinting.” He is now the director of the biomedical engineering program at the University of Texas El Paso. In 2010, Bosworth, a retired executive who previously worked at Dell Computers, met Boland through a mentoring program that matched entrepreneurially minded scientists at the university with business veterans.
“The more I learned about the potential for this technology, the more I thought that this was amazing stuff,” recalls Bosworth. “I said to Thomas one day, ‘we should start a company,’ and he said let’s do it.”
It’s easy to understand Bosworth’s fascination. The technology of 3D bioprinting is at once relatively simple to describe and utterly astounding. Instead of extruding multiple layers of plastic or some other composite to create an inanimate physical object, a bioprinter prints a “bio-ink” of living cells. Typically, layers of different cell types are intermixed with layers of an “extracellular matrix”—a gel in which the cells are suspended.
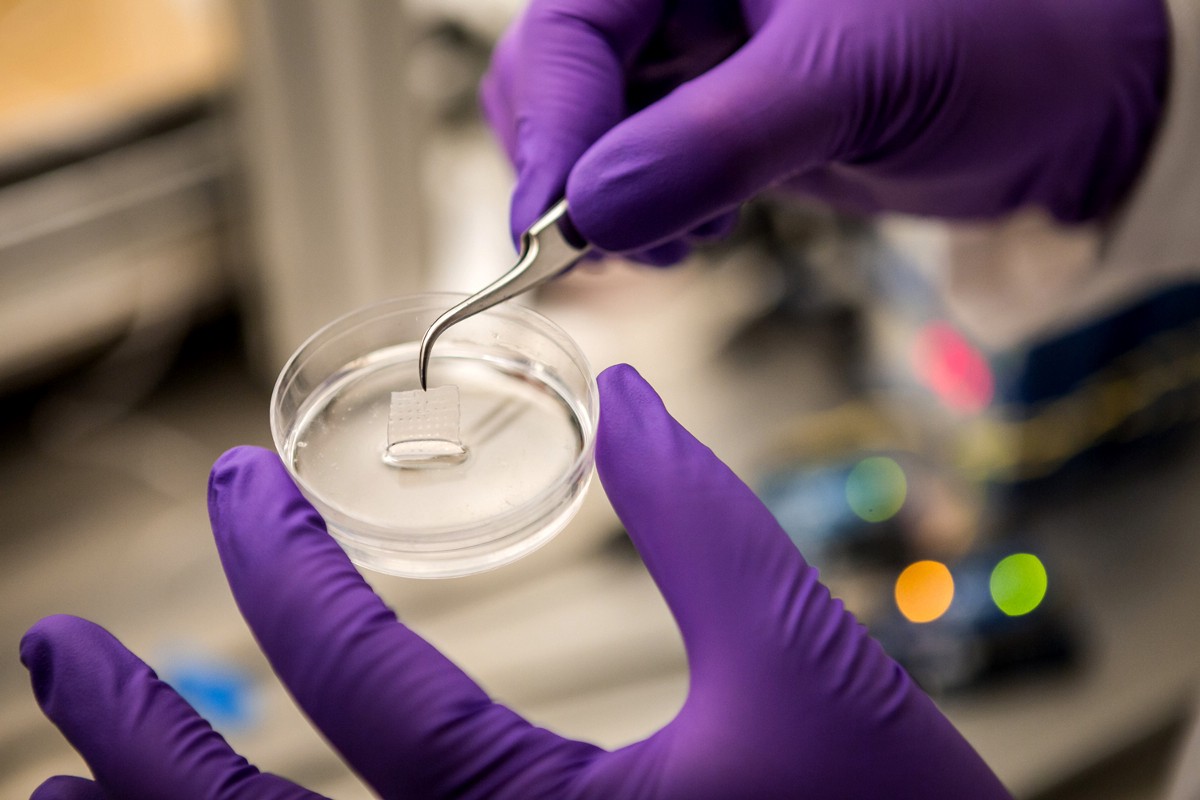 |
| 3D printed tissue construct in a Petri dish (right after printing). Photo courtesy of University of Iowa. |
Cell biologists have been culturing cells and attempting to fabricate larger structures for decades. The advantages delivered by 3D printing, says Boland, are in its precision, flexibility and speed. Different types of cells can be placed in specific locations much more quickly than one could achieve by hand. Speed is of the essence, because the slower the process of assembly, the more likely it is that the cells will die. Using multiple printer heads containing solutions of different cell types and gels, extraordinarily complicated structures can be constructed in short periods of time.
Putting cells in position swiftly is only the beginning. Figuring out how to keep them alive is widely acknowledged as the biggest obstacle scientists face in achieving the holy grail of building fully functional organs that can be transplanted into human bodies.
In the organs in your body, cells are kept alive by nutrients delivered by networks of veins and capillaries—its vasculature. The kidney printed out by Anthony Atala at TED, though shaped like a human kidney, did not have this support network. “Embedding the microvasculature into the printed construct,” says Boland, “has proven very difficult.”
No one, at this point, can say with authority when and how the vasculature problem will be solved. At Harvard’s Wyss Institute, for example, a team led by Jennifer Lewis has won widespread attention for a process in which a branching network of tubes gets printed throughout the extracellular matrix using a bio-ink with a very special property — it melts when it cools. After the full tissue construct is printed, complete with living cells and extracellular matrix and a filament of branching tubes, Lewis’ team chilled the whole thing and sucked out the melted bio-ink, leaving behind an empty network of tubes that, theoretically, can be used to funnel nutrients to the cells.
Boland, who now serves as an advisor to TeVido, says his El Paso lab has been experimenting with printing out “channels” of epithelial cells, the cells that line the walls of blood vessels. The goal is to see if the cells can be induced to self-organize into functional structures, although Scott Collins, TeVido’s chief technology officer, citing intellectual property issues, declined to go into much detail.
“In terms of deliverables, have we made organs yet? No,” Boland says. “But we are getting closer.”
Adds Collins: “we aim to be the first.”
![]()

